Distillation
ENZYMATIC PROCESS FOR BIODIESEL AND BIO-BASED MATERIAL DISTILLATION

Distilled biodiesel is the future of biodiesel. Most of the specifications developed by ASTM or the special quality requirements of large oil companies can be met through biodiesel distillation.
Distilling biodiesel will solve many blending issues with petroleum diesel. It will also eliminate concerns about future ASTM 6751 updates regarding mono-, di-, and tri-glycerides. Additionally, distillation reduces sulfur content (to under 15 PPM) in high-sulfur feedstocks like tallow, chicken fat, or pennycress.
Distillation separates a mixture of two or more components into its purest form. The process involves heating the mixture to a temperature between the boiling points of the components.
For example, at atmospheric pressure, water boils at 212°F, and ethanol boils at about 176°F. If we heat a mixture of water and ethanol to 195°F, the ethanol boils and turns into vapor. This vapor is then collected and condensed, while the water stays as a liquid.
Examples
A fractionating distillation column makes this process more efficient. The picture below shows how columns work.
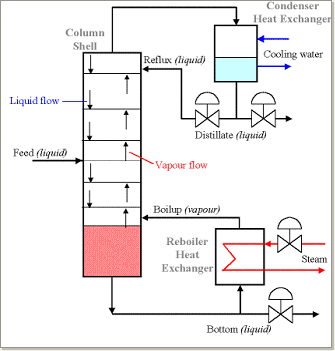
The column consists of stacked plates. A liquid feed enters the column at one or more points. The liquid flows over the plates, and vapor rises through the liquid via holes in the plates. As the liquid moves down the column, the vapor interacts with it multiple times due to the stacked plates. This step is critical in the distillation process. The higher-boiling molecules condense into liquid form. Meanwhile, the lower-boiling molecules turn into vapor, using the energy released.
The base of the column holds a large volume of liquid, mostly made up of the higher boiling liquid (in this case, water). Some of this liquid flows out of the base, is heated in the reboiler, and then returns to the column. This process is called the “boil-up.”

Some vapor escapes from the top of the column and condenses into liquid in the condenser. Part of this liquid returns to the column as reflux, while the rest becomes the top product or distillate. On each plate, the vapor and liquid phases approach thermal pressure and composition equilibrium, depending on the plate’s efficiency.
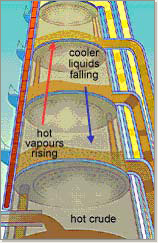
In essence, the system pumps the hot mixture into the bottom of the column. The tower acts as a heat exchanger, removing heat from the rising vapors. As the vapors rise, some condense back into liquid and fall down the column.
The temperature decreases as you move up the column. Different groups of hydrocarbons condense at different heights: the heaviest at the bottom and the lightest at the top. The final product is in its virgin state.
To Read More about SRS’ Distillation Columns and Fractionating Columns Click Here
